ABSTRACT
There is growing concern about the over treatment of cancer because treatments are based primarily on tumor anatomy. In this study, we aim to begin the process of addressing that issue by developing an intravital technique for optically analyzing tumor biology. Two traits were identified as having significant importance in the aggression of a tumor, vascular oxygenation (SO2) and glycolytic demand.
Dorsal skin flap window chambers were implanted and 4T1 and 67NR cancer cells were injected to provide a tumor model for the development of this intravital quantification technique. This study provides a detailed protocol from instrumentation setup to surgical procedures to imaging methods as well as showing that optical techniques can be used to quantify SO2 and glycolytic demand within various cell lines.
METHODS

Figure 2 — The final microscope setup as used in this study with relevant labels.
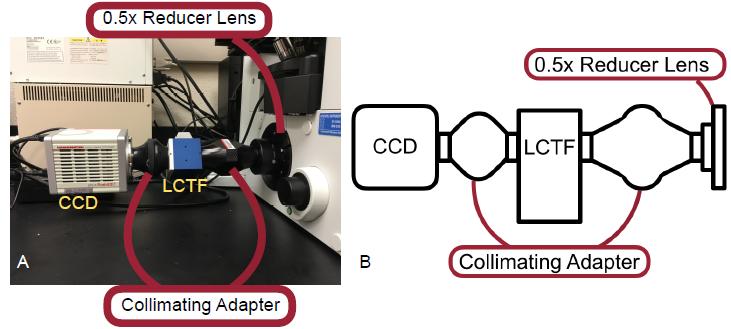
Figure 3 — A. Picture of modular attachments used in this study. B. A schematic of the attachments used.
At its base, the system in use was an motorized inverted fluorescence microscope, the Olympus IX-81. Attached to the microscope was a 100 watt halogen lamp for transmission microscopy, 100 watt mercury lamp used for epifluorescent microscopy, a control box and hand switch, and a sequence of modular attachments to facilitate the unique needs of this study (Figure 2). Those components, pictured below in Figure 3, include a reducer lens, a two-piece collimating adapter, a liquid crystal tunable filter (LCTF), and the CCD.
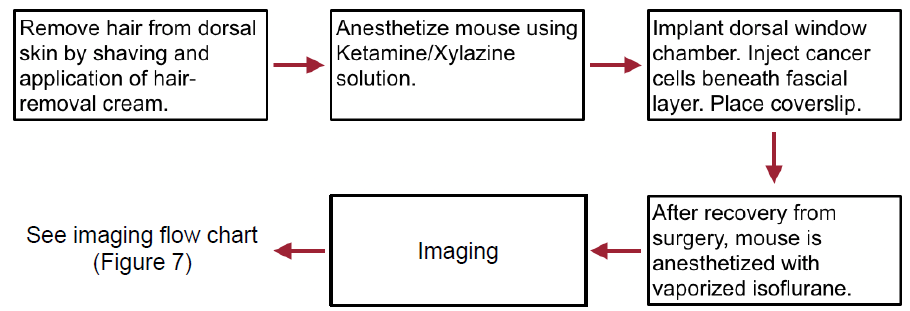
Figure 5 — Flow chart depicting the basic steps of the surgical procedure from preparation to recovery and imaging.
The mice used underwent routine light-dark cycles and were allowed sufficient time for acclimation after arrival to our facility. Additionally, the entire surgical procedure is performed observing sterile techniques. A flow chart of the general procedure is shown in Figure 5.
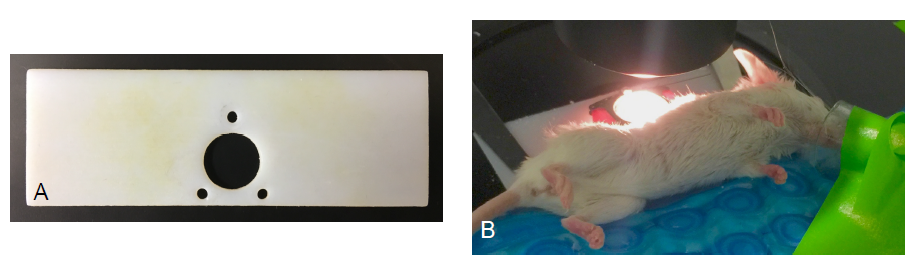
Figure 6 — A. Custom 3D printed imaging platform used to secure mice to stage of microscope. B. Mouse with imaging platform after being secured to microscope and nose cone positioned.
The window chamber was then fitted with a custom, 3D printed imaging platform (Figure 6A) to allow the securing of the mouse and window to the microscope stage. Once secure, an isoflurane nose cone was positioned over the nose of the mouse and used to keep the mouse anesthetized throughout the duration of imaging (Figure 6B). The breathing rate was monitored to ensure no mouse was allowed to overdose on the isoflurane.
Lastly, the data received from imaging would be entered loaded a MATLAB program along with respective ND and dark images for analysis. This entire process is depicted in the flowchart below.
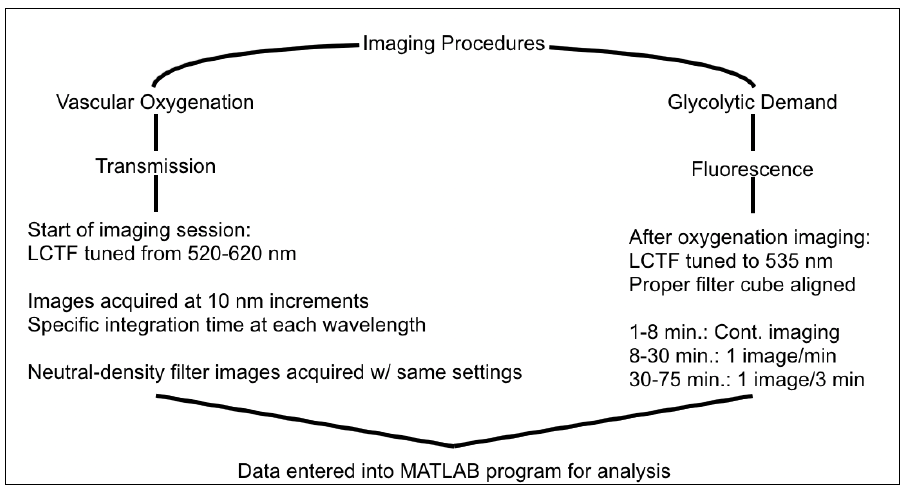
Figure 7 — Flow chart depicting the imaging processes for SO2 and glycolytic demand.
RESULTS AND DISCUSSION
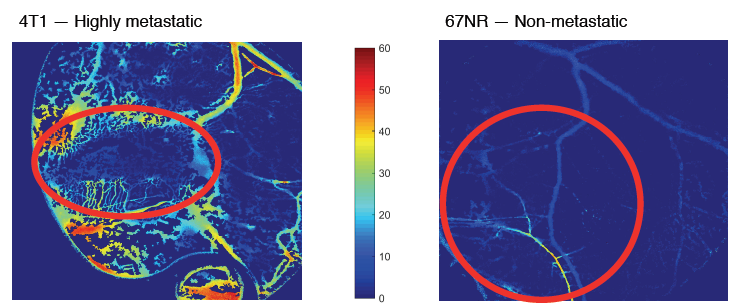
Figure 8 — Transmission images of SO2 with suspicious areas highlighted with a red circle. A. 4T1 tumor 8 days after injection of cells. B. 67NR tumor 15 days after injection.
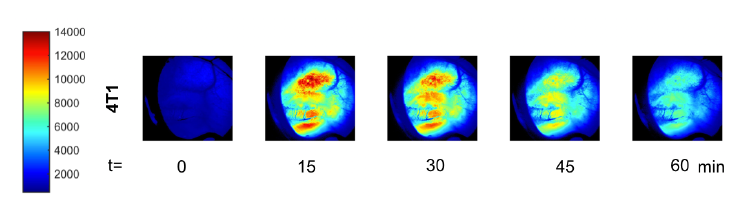
Figure 9 — In vivo uptake of 2-NBDG as quantification of glycolytic demand within 4T1 tumor and surrounding tissue.
In the final six mice at the conclusion and finalization of the protocol, the data showed that it is possible to quantify SO2 and glycolytic demand in vivo. Furthermore, it is clear from the data (Figure 8) that distinctions can be made regarding the vascular oxygenation of different cell lines. Unfortunately, in the collection of glycolytic demand data technical difficulties were experienced in regards to the storage of the data, thus only the 4T1 tumor was able to be analyzed (Figure 9).
CONCLUSIONS
From this study, it is clear that dorsal skin flap window chambers can be used to image and quantify in vivo vascular oxygenation and glycolytic demand. Furthermore, optical techniques can be used to identify the variations in these two phenotypes across cell lines. Additionally, many significant conclusions were drawn about the optimization of the dorsal skin flap window chamber imaging protocol, as discussed above. Over the course of this study, there were several iterations of the protocol, resulting in the refining of the procedures and ultimately, the final protocol. Throughout this refining process, there were several resulting qualitative observations made on the optimal procedures and methods.
Throughout the multitude of surgical window implantations, it was discovered that the surgery should not be performed on any mouse with a mass less than 21 grams. When attempted, these mice showed increased tissue necrosis around the window, minimal tumor growth, and decreased quality of life. This is likely due to the excessive forces involved in stretching the less-abundant skin around the window chamber resulting in increased trauma. Another factor in successful recovery and tumor growth was the nut tightness. Originally, it was expected that the bolts would need to be fastened to firmly hold the two chamber halves together, but this most often resulted in tissue necrosis, as well. If the nuts were loosened in a timely manner, the necrosis could be prevented, though.
Another discovery regarding the nuts and bolts was the importance of directionality. Initially, the skin would be removed on the bolt-protrusion side. This would allow for an increased working distance on the inverted microscope. This increased the difficulty of the saline fill and coverslip and lock ring placement, though. Once the 3D printed platform was doubled in thickness, the added working distance was no longer necessary. Additionally, by changing which side to remove the skin, the future use of two-photon microscopy in the window chambers was made possible, for now the protruding bolts will not hinder the close-proximity objective.
Source: University of Arkansas
Authors: Jesse D. Ivers
>> 200+ Matlab Projects based on Control System for Final Year Students